Crispin Bennet, Heidi Chapman and Laura C Collada Ali of Trilogy Writing & Consulting explore how it’s time for medical device manufacturers to harness the full potential of real-world data to strengthen regulatory submissions and post-market strategies.
The integration of real-world data (RWD) into healthcare decision-making is reshaping how medical devices are developed, assessed and monitored. With RWD becoming increasingly accessible, manufacturers, regulators, and clinicians are recognising its value in enhancing the relevance of research findings, improving patient outcomes, and supporting regulatory decisions.
Medical devices – from in vitro diagnostics to therapeutic technologies – undergo rigorous clinical evaluation before market entry. However, traditional clinical investigations may not fully reflect real-world usage or long-term outcomes, especially for lower-risk devices. In Europe, the Medical Devices Regulation (EU MDR 2017/745) and In Vitro Diagnostics Regulation (IVDR 2017/746) mandate continuous Post-Market Clinical Follow-up (PMCF), requiring manufacturers to collect and assess data throughout the device’s life cycle. Similarly, in the US, regulations such as 21 Code of Federal Regulations Parts 814 and 822 and Section 522 of the Federal Food, Drug and Cosmetic Act establish post-market study and surveillance requirements.
Recent initiatives and publications have focused on leveraging RWD to complement clinical trials and provide deeper insights into device performance, safety, and effectiveness across diverse patient populations. For manufacturers, this means better tools to fill evidence gaps, strengthen post-market surveillance, and guide product development with a more patient-centric approach.
This article reviews three key publications and one collaborative initiative that highlight how RWD is being used in the medical device sector. These resources offer practical insights into regulatory frameworks, data integration methodologies, and real‑world applications that manufacturers should be aware of to optimise their regulatory strategies and product life‑cycle management.
“Manufacturers should explore diverse sources of RWD, including hospital records, electronic health records, registries, and even social media listening.”
Leveraging RWE for legacy devices
As regulatory expectations evolve, RWE is becoming a powerful tool for medical device manufacturers, especially those managing legacy devices. A recent review (McDermott & Kearney, 2024; Expert Review of Medical Devices) highlights how RWE can help bridge gaps in clinical evidence, offering a more accurate and cost‑effective way to demonstrate device safety and performance.
RWD reflects routine clinical practice and typically includes larger, more diverse patient populations than traditional trials. This makes real‑world evidence (RWE) particularly valuable for supplementing pre‑ and post‑market clinical investigations, supporting benefit–risk assessments, and reducing the financial burden of conducting new studies.
Legacy devices—those already on the market but lacking comprehensive clinical data—can benefit significantly from RWE. Regulatory frameworks now offer pathways for manufacturers to use RWD to strengthen clinical evidence, especially under the EU MDR. This approach aligns with initiatives such as the US Food and Drug Administration’s National Evaluation System for Health Technology (NEST), which promotes the use of RWE in regulatory decision‑making.
Manufacturers should explore diverse sources of RWD, including hospital records, electronic health records, registries, and even social media listening. These sources can provide insights into device performance across varied clinical settings, helping to build a robust evidence base.
What are real-world data (RWD) and real-world evidence (RWE)?
RWD refers to observational data collected outside traditional clinical trials – such as electronic health records, insurance claims, and patient-generated data – used to understand patient health and care delivery.
RWE is clinical evidence about the use, benefits, or risks of a medical product, derived from the analysis of RWD.
Defining clinical endpoints using RWD
A novel methodology has emerged that uses RWE to define meaningful endpoints for device evaluation (Wang et al., 2021; Statistics in Medicine). This approach applies entropy balancing, a statistical technique that adjusts for differences between real‑world and study populations. By aligning treatment and control groups more accurately, manufacturers can reduce bias and improve the reliability of conclusions drawn from RWD.
This method is particularly useful when designing clinical investigations or updating performance and safety parameters. It helps manufacturers overcome challenges related to limited or inconsistent data, ensuring that endpoints are relevant and reflective of actual clinical use.
Market access and regulatory submissions
RWE is increasingly recognised by regulators as a valid component of market access strategies. A recent review of the FDA’s framework outlines how RWE can support regulatory approvals, highlighting key legislative milestones and future directions (Shi et al., 2024; Cost Eff and Resour Alloc).
For manufacturers, this means that integrating RWE into submission dossiers can enhance the credibility of clinical evidence and potentially accelerate approval timelines. Understanding how to align RWD collection with regulatory expectations is essential for successful submissions.
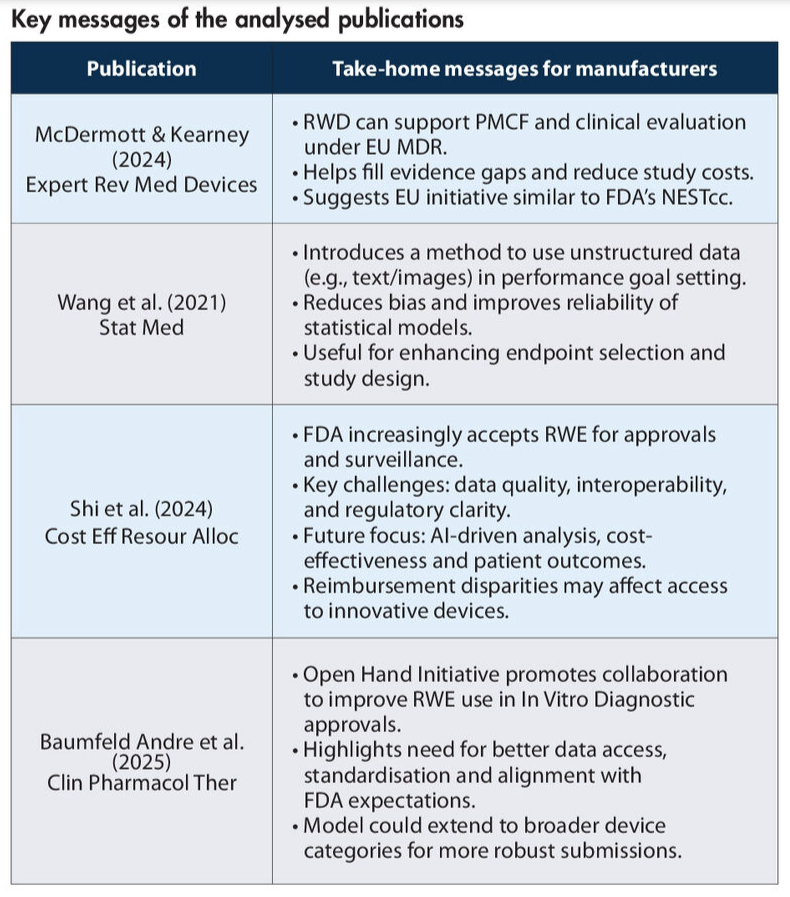
Note: these four publications were identified as most relevant for medical device manufacturers involved in writing regulatory documents for medical devices upon the following search string on PubMed on 24 February 2025: (“real-world data”[Title] OR “real-world evidence”[Title] AND (“medical device”[Title] OR “in-vitro diagnostic”[Title]) AND 2020: 2025[pdat])
Collaborative initiatives: the Open Hand approach
The Open Hand Initiative (Baumfeld Andre et al., 2025; Clinical Pharmacology & Therapeutics), a collaboration between the FDA, manufacturers, and the Medical Device Innovation Consortium, exemplifies how industry and regulators can work together to improve the quality and transparency of RWE. This initiative encourages manufacturers to share insights from regulatory interactions, fostering a more consistent and effective use of RWD in submissions.
Participating in such initiatives can help manufacturers stay ahead of regulatory trends, improve data quality, and ensure that their devices meet evolving standards.
As the medical device industry continues to shift towards evidence-based regulation, RWD offers manufacturers a strategic advantage. Whether supporting legacy devices, defining clinical endpoints, or facilitating market access, RWE provides a cost-effective and reliable way to strengthen clinical evidence.
“Manufacturers should actively engage with emerging methodologies, regulatory frameworks, and collaborative initiatives to ensure their devices are backed by robust, relevant data.”
Conclusion
Manufacturers should actively engage with emerging methodologies, regulatory frameworks, and collaborative initiatives to ensure their devices are backed by robust, relevant data. By doing so, they can enhance patient outcomes, streamline regulatory processes, and contribute to the future of healthcare technology.
Medical writers are uniquely positioned to support medical device manufacturers in navigating these evolving challenges and opportunities, thanks to their deep understanding of regulatory landscapes, clinical evidence generation, and strategic communication.
References available on request.
The content of this article was first published in the European Medical Writers Association Medical Writing Journal in September 2025.
www.medicaldevice-developments.com
https://trilogywriting.com
